The First HIPAA Claims Attachments Standard
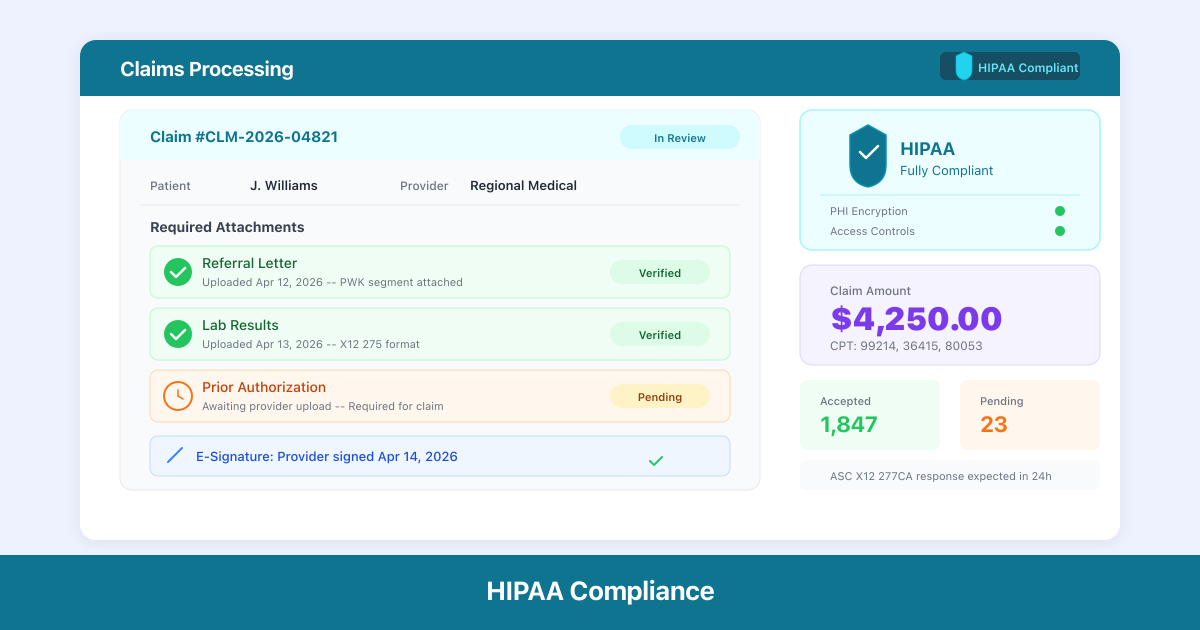
The CMS Final Rule (CMS-0053-F), effective May 26, 2026 with a 24-month compliance window, establishes the first-ever HIPAA-adopted standard for healthcare claims attachments. Providers must now exchange clinical documentation — medical records, imaging reports, lab results, telemedicine notes — electronically with payers using verified electronic signature standards. The rule is projected to save $781 million annually by eliminating fax and mail-based workflows.
What This Means for Healthcare Document Management
Healthcare organizations must implement systems capable of:
- Structured clinical document exchange — replacing fax transmissions with standardized electronic formats
- Verified electronic signatures — authenticating every claims attachment with compliant e-signatures
- Automated attachment routing — matching clinical documents to the correct claims in real-time
- Audit trail preservation — documenting every attachment exchange for compliance verification
Covered entities were also required to update their Notice of Privacy Practices by February 16, 2026 to reflect new reproductive health privacy protections.
UK Companies House: Software-Only Filing from April 2027
In the UK, the Economic Crime and Corporate Transparency Act (ECCTA) 2023 introduces mandatory software-only filing for all company accounts from April 1, 2027 — ending both paper and web portal submissions. Identity verification for all directors became mandatory from November 18, 2025, and all filing agents must be registered as Authorised Corporate Service Providers (ACSPs) by November 2026.
The CLOUD Act vs. Data Sovereignty Tension
The 2026 US National Trade Estimate Report added new sections targeting foreign data sovereignty laws as trade barriers. Meanwhile, the CLOUD Act continues to allow US government access to data stored by US-based providers regardless of physical location. This tension directly affects document management decisions for multinational businesses choosing between US and non-US cloud providers.